
Insights into mechanisms of coccolithophore speciation: How useful is cell size in delineating species?
 based on reviews by Andrej Spiridonov and 1 anonymous reviewer
based on reviews by Andrej Spiridonov and 1 anonymous reviewer
Morphometric changes in two Late Cretaceous calcareous nannofossil lineages support diversification fueled by long-term climate change
Abstract
Recommendation: posted 19 October 2022, validated 25 October 2022
Jarochowska, E. (2022) Insights into mechanisms of coccolithophore speciation: How useful is cell size in delineating species?. Peer Community in Paleontology, 100011. https://doi.org/10.24072/pci.paleo.100011
Recommendation
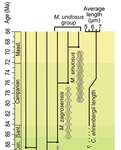
Calcareous plankton gives us perhaps the most complete record of microevolutionary changes in the fossil record (e.g. Tong et al., 2018; Weinkauf et al., 2019), but this opportunity is not exploited enough, as it requires meticulous work in documenting assemblage-level variation through time. Especially in organisms such as coccolithophores, understanding the meaning of secular trends in morphology warrants an understanding of the functional biology and ecology of these organisms. Razmjooei and Thibault (2022) achieve this in their painstaking analysis of two coccolithophore lineages, Cribrosphaerella ehrenbergii and Microrhabdulus, in the Late Cretaceous of Iran. They propose two episodes of morphological change. The first one, starting around 76 Ma in the late Campanian, is marked by a sudden shift towards larger sizes of C. ehrenbergii and the appearance of a new species M. zagrosensis from M. undulatus. The second episode around 69 Ma (Maastrichtian) is inferred from a gradual size increase and morphological changes leading to probably anagenetic speciation of M. sinuosus n.sp.
The study remarkably analyzed the entire distributions of coccolith length and rod width, rather than focusing on summary statistics (De Baets et al., in press). This is important, because the range of variation determines the taxon’s evolvability with respect to the considered trait (Love et al., 2022). As the authors discuss, cell size in photosymbiotic unicellular organisms is not subject to the same constraints that will be familiar to researchers working e.g. on mammals (Niklas, 1994; Payne et al., 2009; Smith et al., 2016). Furthermore, temporal changes in size alone cannot be interpreted as evolutionary without knowledge of phenotypic plasticity and environmental clines present in the basin (Aloisi, 2015). The more important is that this study cross-tests size changes with other morphological parameters to examine whether their covariation supports inferred speciation events. The article addresses as well the effects of varying sedimentation rates (Hohmann, 2021) by, somewhat implicitly, correcting for the stratophenetic trend using an age-depth model and accounting for a hiatus. Such multifaceted approach as applied in this work is fundamental to unlock the dynamics of speciation offered by the microfossil record.
The study highlights also the link between shifts in size and diversity. Klug et al. (2015) have previously demonstrated that these two variables are related, as higher diversity is more likely to lead to extreme values of morphological traits, but this study suggests that the relationship is more intertwined: environmentally-driven rise in morphological variability (and thus in size) can lead to diversification. It is a fantastic illustration of the complexity of morphological evolution that, if it can be evaluated in terms of mechanisms, provides an insight into the dynamics of speciation.
References
Aloisi, G. (2015). Covariation of metabolic rates and cell size in coccolithophores. Biogeosciences, 12(15), 4665–4692. doi: 10.5194/bg-12-4665-2015
De Baets, K., Jarochowska, E., Buchwald, S. Z., Klug, C., and Korn, D. (In Press). Lithology controls ammonoid size distribution. Palaios.
Hohmann, N. (2021). Incorporating information on varying sedimentation rates into palaeontological analyses. PALAIOS, 36(2), 53–67. doi: 10.2110/palo.2020.038
Klug, C., De Baets, K., Kröger, B., Bell, M. A., Korn, D., and Payne, J. L. (2015). Normal giants? Temporal and latitudinal shifts of Palaeozoic marine invertebrate gigantism and global change. Lethaia, 48(2), 267–288. doi: 10.1111/let.12104
Love, A. C., Grabowski, M., Houle, D., Liow, L. H., Porto, A., Tsuboi, M., Voje, K.L., and Hunt, G. (2022). Evolvability in the fossil record. Paleobiology, 48(2), 186–209. doi: 10.1017/pab.2021.36
Niklas, K. J. (1994). Plant allometry: The scaling of form and process. Chicago: University of Chicago Press.
Payne, J. L., Boyer, A. G., Brown, J. H., Finnegan, S., Kowalewski, M., Krause, R. A., Lyons, S.K., McClain, C.R., McShea, D.W., Novack-Gottshall, P.M., Smith, F.A., Stempien, J.A., and Wang, S. C. (2009). Two-phase increase in the maximum size of life over 3.5 billion years reflects biological innovation and environmental opportunity. Proceedings of the National Academy of Sciences, 106(1), 24–27. doi: 10.1073/pnas.0806314106
Razmjooei, M. J., and Thibault, N. (2022). Morphometric changes in two Late Cretaceous calcareous nannofossil lineages support diversification fueled by long-term climate change. PaleorXiv, nfyc9, ver. 4, peer-reviewed by PCI Paleo. doi: 10.31233/osf.io/nfyc9
Smith, F. A., Payne, J. L., Heim, N. A., Balk, M. A., Finnegan, S., Kowalewski, M., Lyons, S.K., McClain, C.R., McShea, D.W., Novack-Gottshall, P.M., Anich, P.S., and Wang, S. C. (2016). Body size evolution across the Geozoic. Annual Review of Earth and Planetary Sciences, 44(1), 523–553. doi: 10.1146/annurev-earth-060115-012147
Tong, S., Gao, K., and Hutchins, D. A. (2018). Adaptive evolution in the coccolithophore Gephyrocapsa oceanica following 1,000 generations of selection under elevated CO2. Global Change Biology, 24(7), 3055–3064. doi: 10.1111/gcb.14065
Weinkauf, M. F. G., Bonitz, F. G. W., Martini, R., and Kučera, M. (2019). An extinction event in planktonic Foraminifera preceded by stabilizing selection. PLOS ONE, 14(10), e0223490. doi: 10.1371/journal.pone.0223490
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article. The authors declared that they comply with the PCI rule of having no financial conflicts of interest in relation to the content of the article.
Evaluation round #2
DOI or URL of the preprint: https://doi.org/10.31233/osf.io/nfyc9
Version of the preprint: 2
Author's Reply, 08 Oct 2022
Dear Recommender,
Dr. Emilia Jarochowska,
We corrected the taxonomic names in figures (4, 6, 9, 10) and modified the minor grammar issues of the text.
Please find the attached files ''manuscript with Tracked Changes''.
Best
MJ.Razmjooei and N.Thibault
Decision by Emilia Jarochowska , posted 01 Oct 2022
, posted 01 Oct 2022
Dear authors,
thank you for your careful and thorough revision, which addresses the points raised by the reviewers. I would like to ask you to correct the taxonomic names in figures (4, 6, 9, 10) to incorporate the new species names you introduce in the text. You have changes sp. nov. 1 and 2 to M. zagrosensis and M. sinuosus throughout the text, but not in these figures; this will be confusing for the readers.
Besides, the text contains a few minor grammar issues. Please have a quick read in the "no markup" mode as they might be harder to pick up with the extensive changes visible, as in the copy I received. These issues concern e.g. the agreement between the subject and the verb in the sentence and a few instances of punctuation, they should be very easy to correct but correcting them will increase the readability of the text.
Thank you for your thorough work and I am very much looking forward to recommending this work and seeing it published.
Kind regards,
Emilia Jarochowska
Evaluation round #1
DOI or URL of the preprint: 10.31233/osf.io/nfyc9
Author's Reply, 09 Sep 2022
Dear Recommender,
Dr. Emilia Jarochowska,
We declare that we have considered all the comments and made almost all corrections as asked by the reviewers.
Please find the attached files ''cover Letter with the response to the reviewers'' and ''manuscript with Tracked Changes''.
Best
MJ.Razmjooei and N.Thibault
Decision by Emilia Jarochowska , posted 26 Oct 2020
, posted 26 Oct 2020
Dear Authors,
I am sorry to get back to you after such a long time. I approached no fewer than 17 reviewers and finally collected two by specialists dealing with coccoliths specifically, as well as size trends in phytoplankton more generally. Both reviewers agree on the importance of this research approach and the material you present, but ask for clarifications concerning a few aspects. The anonymous reviewer asked for a clarification of the aim of the paper. The taxonomic contribution is certainly achieved as the manuscript contains high-quality illustrations and new taxonomic descriptions and observations. However, the study design, the sample size, the paucity of statistical analyses, and the specificity of factors driving the size of single cell organisms limit the last of the aims, i.e. "an illustrative test for the potential link between Cretaceous cooling and the rise in diversity". Both reviewers commented on this latter aim and I second their concerns. Both Cope's and Bergmann's rules have been originally proposed for multicellular endotherms and the application of Bergmann's rule to ectotherms remains debated. In unicellular organisms, "hard" limits on the size (for any given shape) exist, imposed by the rate of intracellular transport, gas diffusion through cell surface and in the cytoplasm, and, for phytoplankton, light acquisition and penetration. A further important component is the structure of the community and the top-down control by grazers. See e.g.
Marañón, E. (2015). Cell size as a key determinant of phytoplankton metabolism and community structure. Annual Review of Marine Science 2015 7:1, 241-264 Cermeño, P., Marañón, E., Harbour, D., & Harris, R. P. (2006). Invariant scaling of phytoplankton abundance and cell size in contrasting marine environments. Ecology letters, 9(11), 1210-1215. Finkel, Z. V., Beardall, J., Flynn, K. J., Quigg, A., Rees, T. A. V., & Raven, J. A. (2010). Phytoplankton in a changing world: cell size and elemental stoichiometry. Journal of plankton research, 32(1), 119-137. Peter, K. H., & Sommer, U. (2012). Phytoplankton cell size: intra-and interspecific effects of warming and grazing. PloS one, 7(11), e49632. and many others
These factors have been extensively documented in extant phytoplankton and, in order to assess the role of cooling in this study, they would need to be addressed with an evaluation of their plausibility, e.g. to evaluate the link between cooling and the rise in diversity, one would need to do a time-series analysis of temperature data and diversity, which is clearly beyond the scope of this, equally valuable, contribution. The study design does not allow to answer this difficult question fully - it definitely merits a discussion, but I am not sure if the data and analyses presented here warrant a definite answer. Both reviewers also requested that the Authors consider whether the observed changes must reflect a global trend or could they be constrained to the given ecosystem? I also agree with the anonymous reviewer that the analyses should be documented in more details. For example, the confidence intervals provided in tables and figures were calculated based on the assumption of the normality of their distributions (which is warranted, looking at the histograms), but it does not seem to be stated anywhere. What is the white dashed line in Fig. 3, which highlights the shift in length, but must be clearly a running average, because it does not follow all small-scale shifts before and after the highlighted interval? It should be documented how it was calculated and, less importantly, individual fragments of the figure should be labeled and referred to individually in the text to make it easier for the reader to match the described results with presented data. I also struggled with the lack of labels for symbols in Fig. 10 - it is hard to assess to assess the relationship between variables without this information.
Finally, the anonymous reviewer also requested clarifications concerning preservation of the fossils.
In my opinion, the strength of this article is the excellent documentation of an important contribution to nannofossil diversity, high-quality taxonomic data and illustrations, but the link with Cope's and Bergmann's rule warrants a bit more nuanced discussion. Apart from this, this is without doubt an important and well-documented contribution. I would also like to highlight that the authors provided a complete set of data used for the study in an open access archive, which is highly commendable! I apologize again for the very long time it took to solicit reviews and look forward to the revised version.
Kind regards, Emilia Jarochowska
Reviewed by Andrej Spiridonov, 02 Sep 2020
This is very interesting morphometric stratophenetic study of an important Cretaceous algae group in the understudied Iran region. The basic size spectrum patterns are clearly detected and described. The only thing that should be improved is the ultimate explanation for the found trends, which need a conceptual improvement. My comments are below:
In the abstract it is mentioned that two species were studied, but afterwards it is explained that two more are distinguished. I think that you should list four species studied, two of which newly distinguished in this contribution.
Line 45 “…morphospecies species” -> “morphospecies” since redundant.
Figure 1. A) lacks paleogeographical legend, therefore it is difficult to interpret. What Sh means? Shahneshin section (it should be noted in the legend)? Is it site of the study? Why it is on (apparent) paleo-land (?) and not under paleo-water if I understand the map correctly? Probably paleogeography of the studied interval should be significantly different then.
Figure 10. What those colors of data points indicate? Different data sources (studied sections)? Those should be indicated in the legend or the description of the figure.
The main point I see in the article is the explanation of size trends. Authors propose that the increases in sizes of nannoplankton which are associated with profound climate cooling events are caused by Cope and Bergman rules. Contrary to the authors which see solutions here, I see a paradox here. Bergman rule which states, that in colder temperatures body size increases mostly works for endotherms such as mammals, which body size increase diminishes surface area to volume ratio, thus saving the energy and resources. In phytoplankton world rather different rules apply since radical differences in bioenergetics, life history and dominant physical forces at different size scales.
Based on the first principles of geometry, knowledge of eukaryote metabolism, modern observations of the phytoplankton, and climatology we should expect to have positive correlation of cell size increase with the average temperature. This follows from the fact that increased temperature causes higher saturation of atmosphere with water vapour and general increase in intensity of hydrological cycle and thus the flux of nutrient from continents to the oceans. Smaller cells have larger relative surface area, and relatively smaller inner metabolic volume, which allows them to thrive in nutrient poorer environments. Larger cells (all else being equal) require higher density of nutrients which could be diffusively intaken and used by disproportionately larger volume of biochemical machinery. Exactly this pattern was found by our team when studying lundgreni event in the Silurian (Spiridonov et al, 2016 - http://www.geology.cz/bulletin/contents/art1679) – during cooling there was sharp decrease in algae cyst sizes, and during subsequent warming there was a steady growth in their sizes.
Therefore instead of citing broad principles of questionable applicability for the given case I suggest for authors to rethink the probable causality of their excellently documented morphometric and diversity pattern. The association of cooling with increase in size (and diversity?) is against the expectation (and other empirical evidence from differing time periods) for a given group of organisms, and it is an interesting observation which could give some clues on the deeper understanding of phytoplankton ecosystem paleoecology.
My suggestions for the improvement of Discussion:
-Copes rule possibly played a role here (in a style of Steven Stanley/S.J. Gould explanation of release of a clade after extinction event). If for example other competing groups were disproportionately eliminated from the competitive pool, possibly good portion of previously unavailable nutrients were relocated to the calcareous nannoplankton which could have played a role in size increase,
-Bergman rule is too speculative here, and vaguely grounded elsewhere, and probably should be abandoned as a general source of explanation. Instead authors should think deeper what factors which are associated with this cooling episode counteracted the size decreasing effects of cooling which are expected from the first principles. Possible sources of size increase: i) cooling induced increased dust flux due to aridization from deserts to the oceans; ii) mentioned restructuring of competition/grazing networks in the ocean; iii) specifics of physiology/shape/ecology of given clade of algae; iv) other sources of nutrient enrichment and simultaneous cooling – underwater igneous activity, e.g. in Caribbean Igneous Province (???).
Sincerely,
Prof. Andrej Spiridonov Department of Geology and Mineralogy Vilnius University
https://doi.org/10.24072/pci.paleo.100047.rev11









